
Ideal Medical Technologies to Present at Investor Meetings September 16, 2024|Investor Update IMT has been chosen to present at the NC MedTech Venture Day forum on September 19th, which will be held at the Raleigh Convention Center. IMT has also been chosen to present at the BioFuture 2024 investors meeting to be held in New York, from October 28-30th. 
Ideal Medical Technologies Expands its Simulation Capabilities August 1, 2024|Simulator Update Jeremy DeJournett, IMT's Chief Technical Officer, expanded the simulation platform we use to optimize the FUSION systems glucose control software. This new simulation platform, which is informed by data from published ICU studies, is capable of performing 100K five -day "ICU Patient" simulations per day. The results from these simulations are automatically generated and allow us to compare different versions of our glucose control software. We can also look at individual patient simulations, which helps us to further optimize our glucose control software. Additionally, this new platform simultaneously checks for software bugs, which helps us to improve the overall safety profile of the FUSION system. These simulation capabilities help us to optimize the performance of the FUSION systems glucose control software and improve its safety. 
Ideal Medical Technologies Updates its Software Documentation to Remain Compliant with new FDA Standards June 19, 2024|Software Update In June of 2023, the FDA released a new guidance document pertaining to software documentation for medical devices. In order to remain compliant with these new requirements, IMT worked with the software experts at our chosen original equipment manufacturer, Velentium, to make sure the FUSION system software documentation would adhere to the new FDA standards. 
Ideal Medical Technologies Completes it's First-In Human Safety Study of The FUSION system March 13, 2023|Clinical Study IMT successfully completed it's first-in-human clinical study of the FUSION system. The seven study participants, who had type 1 and 2 diabetes, ate three meals over a 24-hour period while the FUSION system attempted to control their glucose to a range of 100-140 mg/dL. The meals were unannounced to the FUSION system. For this study, the FUSION system achieved an average percent in range 70-180 mg/dL of 87.8%, and an average percent time in range less than 70mg/dL of 0.2% and a coefficient of variation of 19.4%. These results were presented at the 2023 American Diabetes Association meeting. 
Ideal Medical Technologies Completes it's First-In-Human Safety Study of the FUSION System
In May 2023 IMT successfully completed the first-in-human clinical study of the FUSION system. In this unannounced 24 hour meal challenge study, the FUSION system achieved excellent safety and efficacy results. These results included an average percent time in range 70-180mg/dL of 87.8%, an adverage percent time in range less than 70mg/dL of 0.2%, and a coefficient of variation of 19.4%. These results were presented at the 2023 American Diabetes Association meeting. 
IMT successfully completed its first in human clinical study of the FUSION system. The study participants, who had type 2 diabetes, ate three meals over a 24-hour period while the FUSION system attempted to control their glucose to a range of 100-140 mg/dL. For this study, the FUSION system achieved an average percent time in range 70-180 mg/dL of 97.3%, and allowed no values less than 70 mg/dL.

June 1, 2022|Financial The NIDDK division of the NIH has awarded IMT a $1.9M SBIR grant. This non-dilutive grant will be used to cover the costs for ongoing development of the commercial grade FUSION system and both safety and randomized studies of the FUSION system in the ICU setting. 
IDE Application for Early Feasibility Study March 24, 2021 | Clinical Study, Regulatory The FDA has approved IMT's investigational device exemption (IDE) application for our first in human study of the FUSION system. This IDE approval will allow IMT to file a series of supplemental IDE expansion requests with the FDA in the future, in order to obtain regulatory approval for our planned future clinical studies. Supplemental requests are easier to prepare, which will shorten the time interval between our future studies. 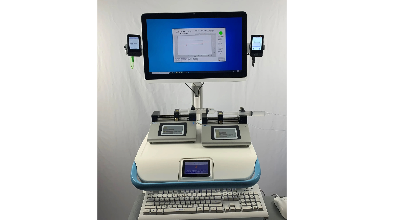
FUSION Artificial Pancreas System January 25, 2021|Business Development, The prototype of our fully autonomous FUSION artificial pancreas system is pictured here. This prototype system, which will be used in our original clinical studies, consists of two Dexcom G6 continuous glucose monitors, our AI based glucose control software running on a Medical Computer (Teguar), and two syringe pumps. The entire system is mounted on a powered Medical Cart (Enovate). The pictured Medical Computer and Medical Cart have already been approved for use in the hospital setting. The FDA is allowing use of the Dexcom G6 CGM system to continuously monitor glucose levels of COVID-19 patients in the ICU setting. 
Breakthrough Medical Device Designation
March 26, 2019|Regulatory
In March of 2019, the Food and Drug Administration (FDA) classified Ideal Medical Technologies FUSION system as a Breakthrough Medical Device. The FDA's Breakthrough Medical Device program is designed to expedite patient access to breakthrough technologies that have the potential to offer more effective treatment for life-threatening conditions, such as poor glucose control in the ICU setting. By having this designation, we are able to get feedback from the FDA on issues involving our FUSION system within 30 days, versus the usual 75-90 day wait experienced by other companies trying to bring medical devices to market, that have not been designated as breakthrough devices. This quicker FDA feedback will help to shorten the time it takes to bring the FUSION system to market as a therapy for COVID-19
|